February 2026
The TRANSFORM study evaluated Breyanzi, an autologous CD19-directed CAR T-cell therapy, versus standard of care (SOC) as second-line treatment in adults with relapsed or refractory large B-cell lymphoma (LBCL). Patients were primary refractory to or relapsed within 12 months of first-line therapy and were eligible for autologous stem cell transplantation.
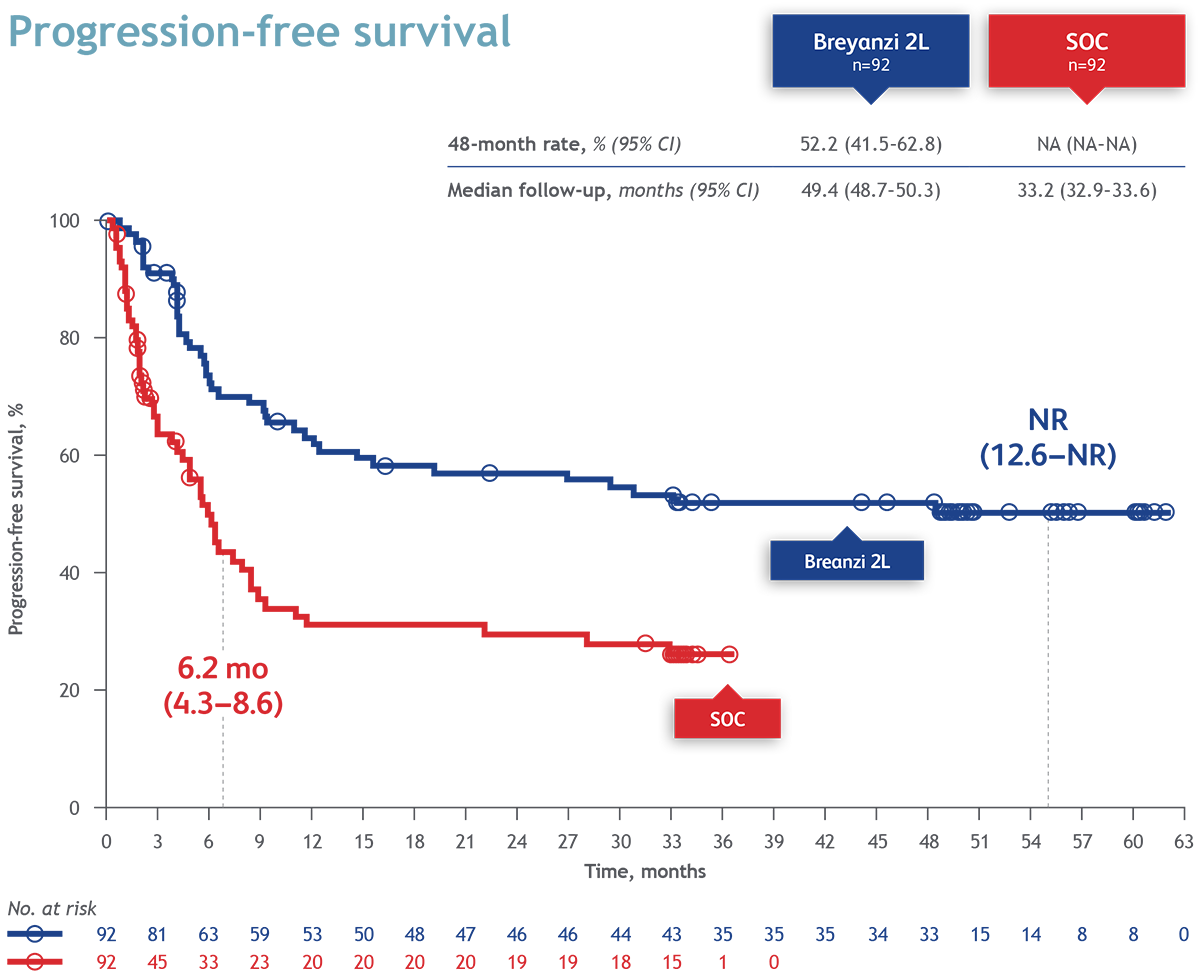
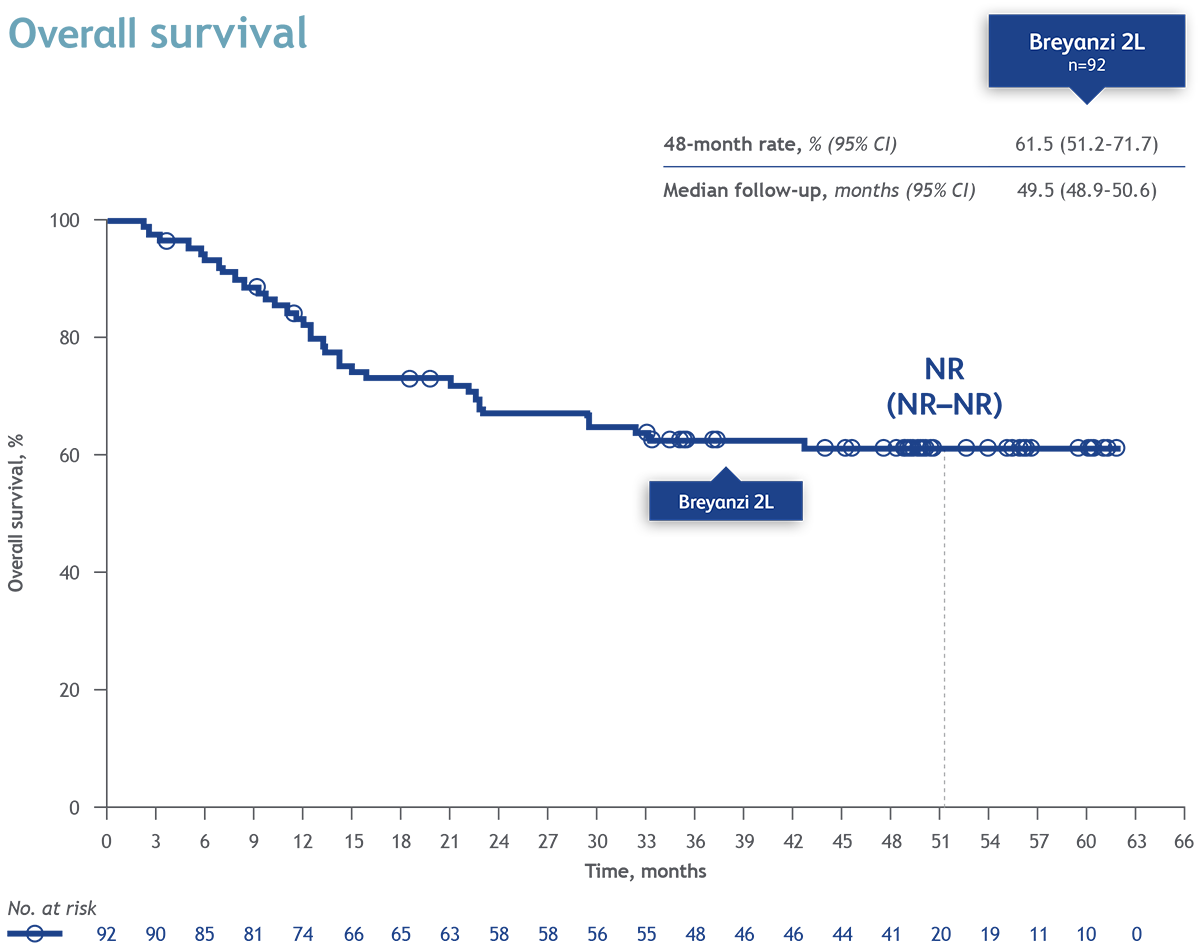
After completion of TRANSFORM, patients treated with Breyanzi could enroll in a long term follow-up (LTFU) study, with a combined median follow-up of approximately 4 years. In patients enrolled in the LTFU study the median Progression-Free-Survival (PFS) was 52.5% (95% CI 41.5–62.8) and overall survival (OS) 61.5% (95% CI 51.2–71.7), at 48 months. In patients from the LTFU study in the Breyanzi arm (n=43), median PFS and OS were both not reached (NR) with 95% CIs of NR–NR. The safety results of Breyanzi in this LTFU study showed no new signals compared with previous reports from TRANSFORM.
Conclusion: After a median follow-up of approximately 4 years, combining data from the TRANSFORM and LTFU studies, Breyanzi continued to demonstrate long-term clinical benefit with stable PFS and OS rates and a manageble safety profile. These results support Breyanzi as a second line treatment, with curative potential, in patients with R/R LBCL.