February 2026
Breyanzi was evaluated in the TRANSCEND NHL 001 study in patients with relapsed or refractory mantle cell lymphoma (MCL) who had received ≥ two lines of prior systemic therapy, including a BTK inhibitor. The study population was heavily pretreated and included patients with high-risk disease characteristics.
Results: Breyanzi demonstrated high and rapid response rates, with an overall response rate (ORR) of 83% (95% CI, 73.3–90.5) and complete response (CR) rate was 72.3% (95% CI, 61.4–81.6). Median time to first CR or PR was 0.95 months (range 0.7–3.0). The safety profile was manageable, with low rates of severe cytokine release syndrome and neurologic events – CRS grade 3/4 = 1% and grade 5 = 0, NE grade 3/4 = 9% and grade 5 = 0.
Study highlights
- High efficacy: Overall response rate of 83.1%, with 72.3% of patients achieving a complete response
- Consistent responses across high-risk subgroups, including patients with TP53 mutations, blastoid morphology, and secondary CNS involvement
- Manageable safety profile, with low incidence of grade ≥3 cytokine release syndrome and neurologic events
- Rapid onset of response, with most patients responding within the first month after infusion
Response rate
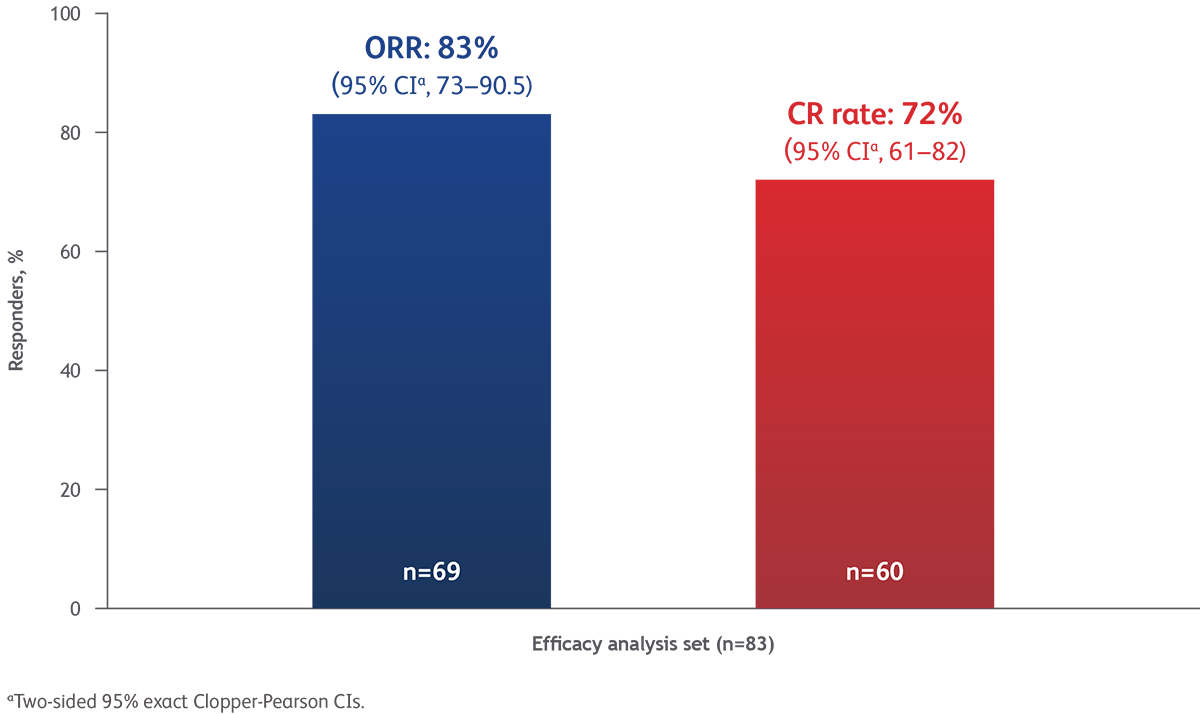
Conclusion: These results demonstrate that Breyanzi provides rapid, deep, and durable responses with a favorable benefit-risk profile in heavily pretreated patients with relapsed or refractory mantle cell lymphoma, supporting its role as a treatment option after ≥ two lines of prior systemic therapy, for patients with limited alternatives after BTK inhibitor failure.

